Test system definition
Use Test system definition documents to define a collection of suitability tests and corresponding margins for quantitative response assays.
About this document type
Test system definition documents let you set up a centralized suitability test system for repeated use. Instead of defining tests within each Quantitative response assay document, you reference a shared Test system definition document to ensure consistency across your assays.
You can include one or more suitability tests in each Test system definition document and define their parameters, scope, and severity level. For details on these settings, see the Suitability testing topic.

To set up these references, you use the Content editor of your Quantitative response assay document. For details on the required steps, see the Create references to documents topic.
Document structure
Each Test system definition document have one default section, that is Suitability tests, plus one or more optional Comment sections. Use the subsections to add the assay, sample, and document suitability tests you require.
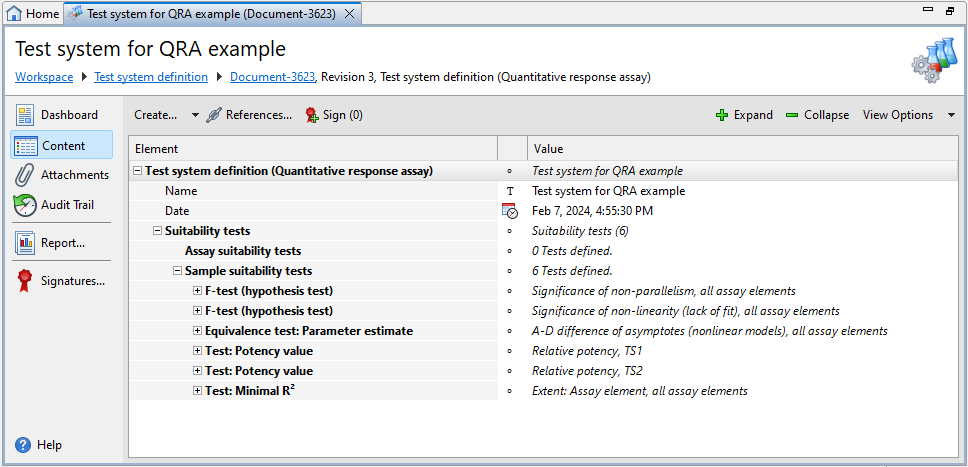
Suitability tests
| Type | Description |
|---|---|
|
Assay suitability tests |
The results of assay suitability tests affect the whole assay document rather than just individual Standard-vs-Test sample comparisons. |
|
Sample suitability tests |
The results of sample suitability tests just affect the individual Standard-vs-Test sample comparison for which they are set rather than the whole assay document. |
|
Document suitability tests |
The results of document suitability tests affect the whole assay document. |
